Pfizer-BioNTech Seeks Emergency Authorization for Second Booster Shot for Elderly
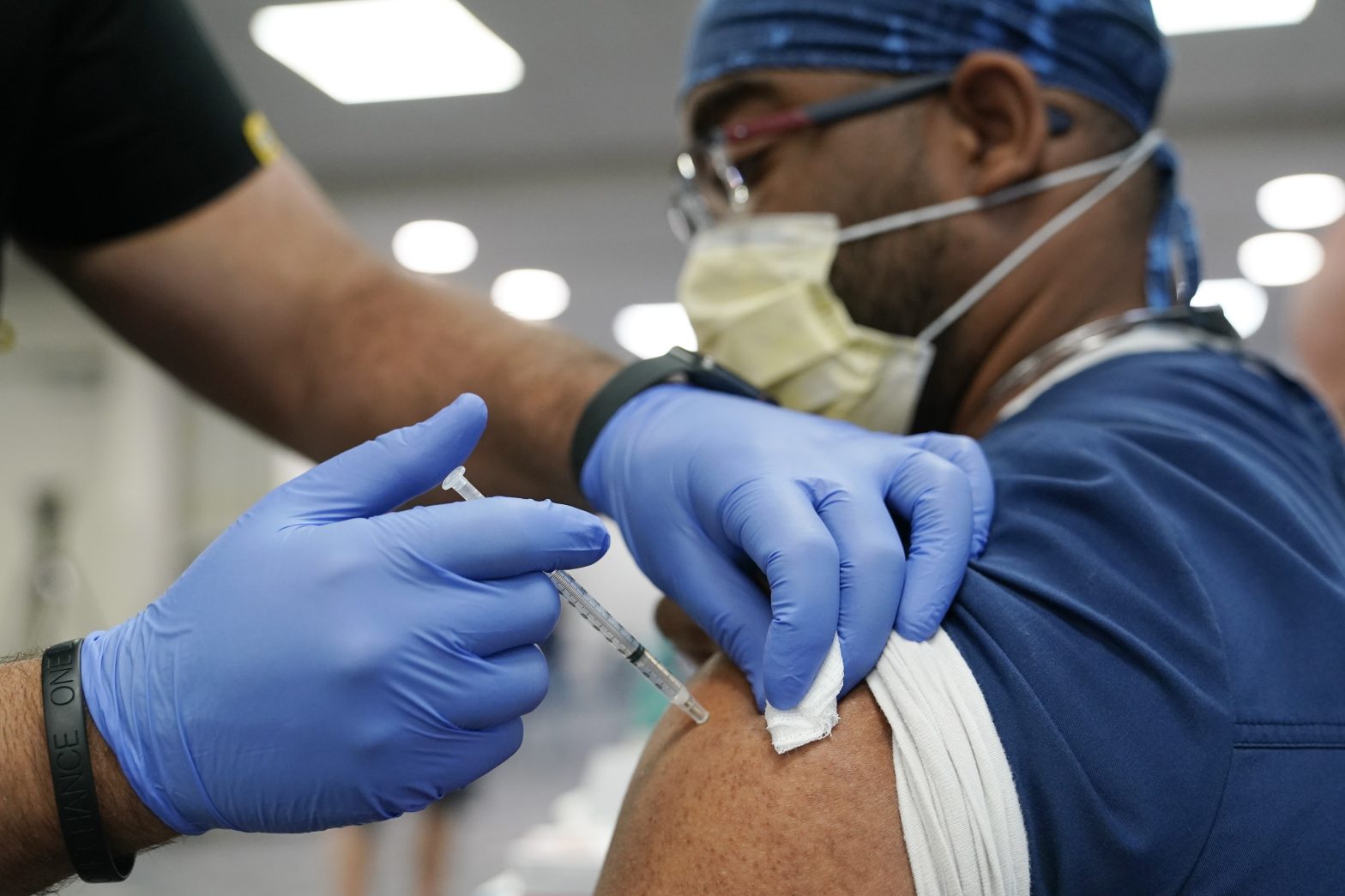
Pfizer-BioNTech announced that they will seek emergency authorization from the U.S. Food and Drug Administration for a second booster shot for adults 65 and older.
The decision to submit an application to the FDA comes after real-world data sets from Israeli researchers showed that an additional booster can increase immunity, lower infection rates and prevent severe illness in adults aged 60 and older.
A study completed by researchers at Kaiser Permanente Southern California suggests that the effectiveness of the third vaccine dose — against both symptomatic COVID-19 infection and severe disease caused by the omicron variant — wanes 3 to 6 months after the individual receives it.
Pfizer-BioNTech advise in their announcement that the data from the KPSC study has been submitted to the FDA as an example of the waning efficacy of the vaccine, but that has not yet been reviewed by the agency.
Alexa can be reached at [email protected]

























