Lawmakers Urge Manufacturers of Naloxone to Apply for Over-the-Counter Status
U.S. Sen. Tammy Baldwin, D-Wis., and Rep. Carolyn B. Maloney, D-N.Y., Chairwoman of the House Committee on Oversight and Reform, along with Sens. Maggie Hassan, D-N.H., and Angus King, I-Maine, and Rep. Brian Fitzpatrick, R-Pa., led a bicameral, bipartisan group of congressional colleagues in letters sent to seven major manufacturers of naloxone urging them to apply for over-the-counter status for their products to help increase access to the lifesaving medication.
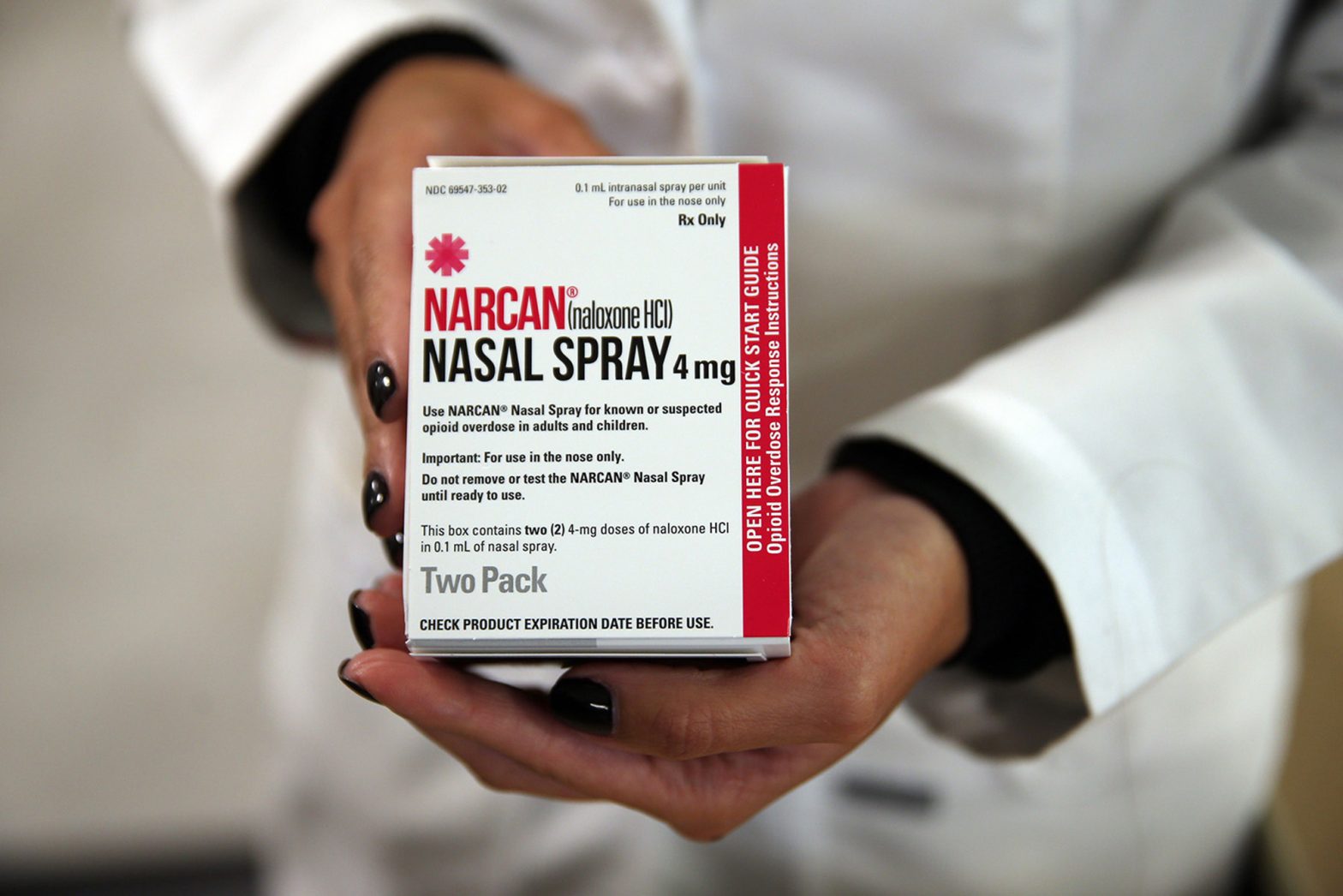
Naloxone is an opioid overdose reversal medication, and evidence has shown its ability to reduce opioid overdose mortality rates by 46%.
The letters from members of Congress were sent to the CEOs of Pfizer, Emergent BioSolutions, Teva, Hikma, Akorn, Adamis and Amphastar Pharmaceuticals.
According to the letters, the only reason naloxone is not available as an over-the-counter treatment is because these drug manufacturers have resisted applying.
In 2019, the product received public support from the U.S. Food and Drug Administration to be made available over-the-counter.
The letters come as the COVID-19 pandemic has exacerbated the substance use disorder crisis in the country and the opioid epidemic.
In 2021, the United States experienced more than 100,000 overdose deaths in a 12-month period for the first time ever.
Alexa can be reached at [email protected]