First US Patient Receives Patient-Derived Implant to Treat a Leading Vision Loss Cause
WASHINGTON — A surgical team at the National Institutes of Health has successfully implanted a patch of tissue made from the patient’s own cells to treat advanced “dry” age-related macular degeneration, a leading cause of vision loss among older Americans.
Currently dry AMD, also known as geographic atrophy, has no known treatment.
In a release, the NIH said the unnamed patient received the therapy as part of a clinical trial that is the first in the United States to use replacement tissues from patient-derived induced pluripotent stem cells.
The surgery, the culmination of 10 years of research and development, was performed by Dr. Amir Kashani, Ph.D., associate professor of ophthalmology at the Wilmer Eye Institute at Johns Hopkins School of Medicine with assistance by Dr. Shilpa Kodati, a staff clinician at the National Eye Institute.
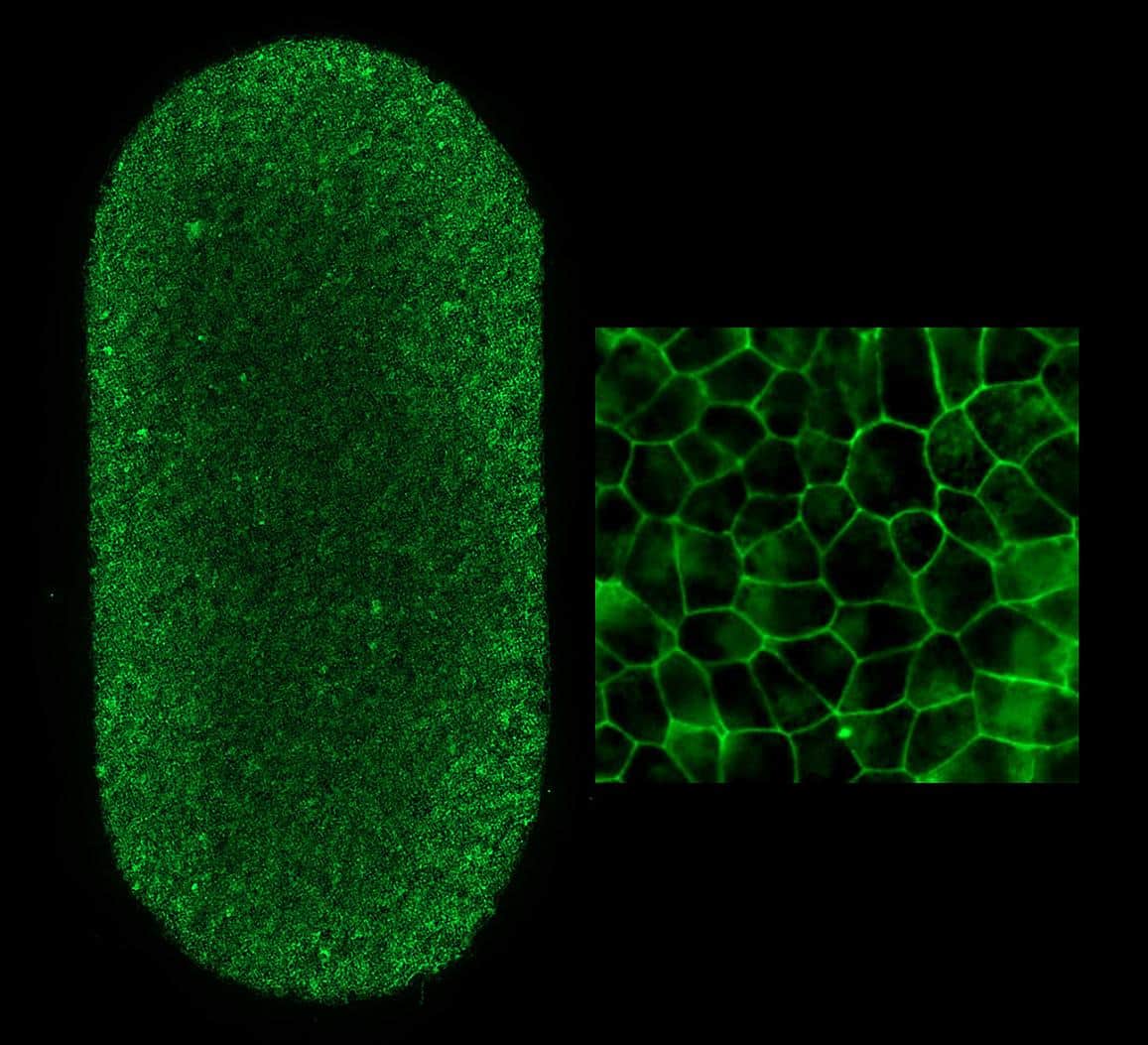
Prior to the procedure, the patient’s blood cells were converted to iPS cells in a National Institutes of Health lab. Once converted to iPS cells, the blood can become almost any type of cell in the body.
In this case, they were programmed to become retinal pigment epithelial cells, the type of cell that degenerates in the advanced forms of dry AMD.
RPE cells nourish and support light-sensing photoreceptors in the retina. In AMD, the loss of RPE leads to the loss of photoreceptors, which causes vision loss.
The procedure was performed at the NIH Clinical Center in Bethesda, Maryland, under a phase 1/2a clinical trial to determine the therapy’s safety.
This iPS cell-derived therapy was developed by the Ocular and Stem Cell Translational Research Section team led by Kapil Bharti, Ph.D., senior investigator at the National Eye Institute, in collaboration with FUJIFILM Cellular Dynamics Inc., and Opsis Therapeutics, based in Madison, Wisconsin.
The safety and efficacy of the cell therapy was tested by the NEI preclinical team. Clinical-grade manufacturing of this cell therapy was performed at the Center for Cellular Engineering, Department of Transfusion Medicine, Clinical Center, at the National Institutes of Health.
This work was supported by the NIH Common Fund and NEI Intramural funding.
Dan can be reached at [email protected] and @DanMcCue
























